[화학공학] PVAc와 PVA 합성(영문)
 등록일 / 수정일
등록일 / 수정일 페이지 / 형식
페이지 / 형식 자료평가
자료평가 구매가격
구매가격
- 2011.11.21 / 2019.12.24
- 24페이지 /
 ppt (파워포인트 2003)
ppt (파워포인트 2003) - 평가한 분이 없습니다. (구매금액의 3%지급)
- 1,800원
최대 20페이지까지 미리보기 서비스를 제공합니다.
자료평가하면 구매금액의 3%지급!
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8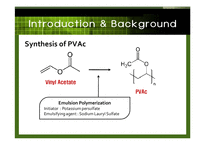 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20
추천 연관자료
- 목차
-
1.Objective
2.Introduction & Background
3.Apparatus & Reagents
4.Procedures
5.Reference
- 본문내용
-
PVAc (Poly vinyl acetate)
A rubbery synthetic polymer
with the formula (C4H6O2)n
A hard, colorless, and nontoxic substance.
An intermediate product in the synthesis of polyvinyl alcohol.
Emulsion Polymerization
starts with an emulsion incorporating water, monomer, and surfactant.
Advantage
- High molecular weight polymers can be made.
- Heat can be removed from the system easily.
Disadvantage
- The polymer is obtained impure.
The process of exchanging the organic group R″ of an ester with the organic group R′ of an alcohol.
These reactions are often catalyzed by the addition of an acid or base catalyst.
Reagents
Methanol
Molecular formula : CH3OH
Molar Mass : 32g/mol
Appearance : Colorless liquid
Density : 0.79g/cm3
Melting point : -97.8℃
Boiling point : 64.7℃
- 참고문헌
-
Chemical Engineering Laboratory Ⅰ/p.25-27
http://joylab.net
http://www.britannica.com/EBchecked/topic/469233/polyvinyl-acetate-PVAc
http://mslab.polymer.pusan.ac.kr/sub4/pva.html
http://en.wikipedia.org/wiki/Vinyl_acetate
http://en.wikipedia.org/wiki/PVAc
http://www.absoluteastronomy.com/topics/Emulsion_polymerization
자료평가
-
아직 평가한 내용이 없습니다.










